Introduction
Autoimmune diseases represent one of modern medicine’s most intricate puzzles—conditions where the immune system, designed to defend, begins to attack the body’s own tissues. Over 80 autoimmune conditions have been identified, including rheumatoid arthritis, lupus, Hashimoto’s thyroiditis, celiac disease, and type 1 diabetes. While each affects different organs, they share one fundamental feature: a breakdown of immune tolerance.
Emerging research reveals a critical player in this breakdown—the intestinal barrier. Once considered a passive wall for nutrient absorption, the gut lining is now recognized as an active immune and metabolic organ. When this barrier becomes compromised—a state often referred to as “leaky gut” or increased intestinal permeability—immune cells encounter dietary proteins, bacterial fragments, and toxins they were never meant to see. This exposure can trigger chronic inflammation, immune confusion, and, over time, autoimmune reactivity.
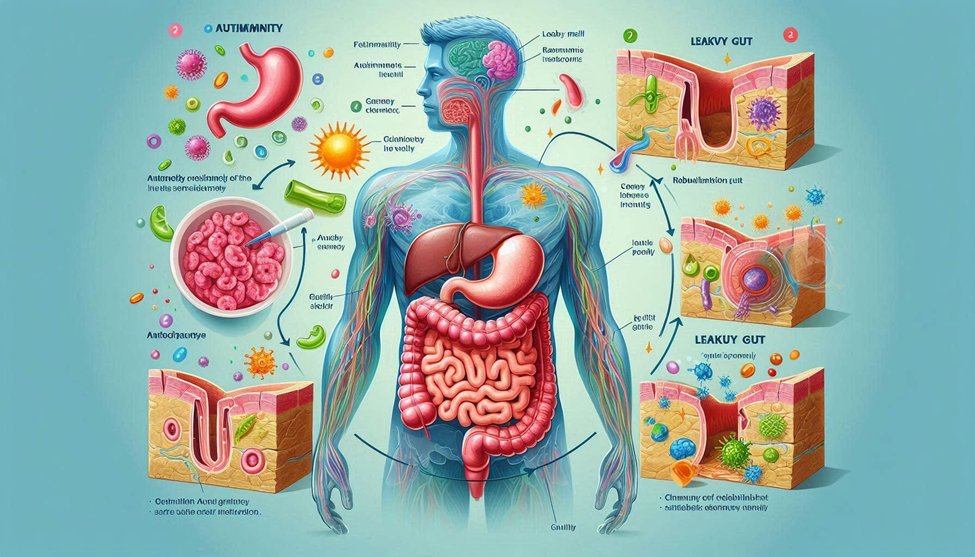
Understanding and repairing the intestinal wall is therefore not just about digestion—it is about immune restoration. The gut is where 70–80% of the immune system resides. When the barrier weakens, it can distort the immune orchestra that maintains self-tolerance. To rebuild the intestinal wall is to restore the physical and biochemical boundaries that distinguish “self” from “non-self.”
1. The Architecture of the Gut Barrier
The intestinal wall is more than a simple lining—it is a multilayered, intelligent structure composed of epithelial cells, tight junction proteins, mucosal secretions, and a dense community of microbes that together regulate what enters the bloodstream.
- Mucus Layer: The first line of defense. Rich in cumin glycoproteins, it traps pathogens and acts as a habitat for beneficial bacteria.
- Epithelial Cells: These cells form a continuous single-cell layer, joined by tight junctions (Claudine, occluding, and zonal occludes). These junctions open and close dynamically in response to diet, stress, and microbial signals.
- Immune Layer: Beneath the epithelium lies the gut-associated lymphoid tissue (GALT), containing immune cells like dendrite cells, macrophages, and lymphocytes that constantly sample antigens.
- Micro biome: Trillions of bacteria regulate barrier integrity by producing short-chain fatty acids (SCFAs), competing with pathogens, and modulating inflammatory cytokines.
When this architecture is disrupted, foreign antigens can “leak” through the gaps between cells, activating immune responses that were never meant to occur.
2. Mechanisms behind “Leaky Gut”
Leaky gut, scientifically termed increased intestinal permeability, occurs when the tight junctions lose selectivity. Factors that compromise this barrier include:
- Dietary antigens: Gluten, processed foods, refined sugars, and emulsifiers can increase permeability by stimulating the protein zonulin, which loosens tight junctions.
- Microbial imbalance (symbiosis): Loss of beneficial bacteria and overgrowth of pathogens trigger inflammation, impairing epithelial repair.
- Chronic stress: Elevates cortical and corticotrophin-releasing hormone (CRH), both of which reduce mucus secretion and weaken junction integrity.
- Medications: NSAIDs, antibiotics, and proton-pump inhibitors (PPIs) are known to damage the intestinal lining.
- Toxins and alcohol: Alcohol metabolites and environmental toxins (like glyph sate and BPA) can thin the mucosal layer and disrupt cellular cohesion.
The result is a “porous” barrier, where bacterial fragments (lip polysaccharides, or LPS) and undigested food particles cross into the bloodstream. The immune system interprets them as invaders, generating chronic low-grade inflammation and the production of auto antibodies that may mistakenly target host tissues.
3. from Leaky Gut to Autoimmunity
The connection between gut permeability and autoimmunity is supported by growing clinical evidence. In genetically susceptible individuals, intestinal barrier dysfunction can precede the onset of autoimmune diseases.
- Celiac disease provides the classic model: gluten triggers zonulin release, tight junctions open, and gliding fragments stimulate immune attack on the intestinal villa.
- Type 1 diabetes has been linked to increased intestinal permeability years before diagnosis, allowing bacterial components to enter circulation and activate pancreatic autoimmunity.
- Hashimoto’s thyroiditis may be exacerbated by cross-reactivity—antibodies against bacterial or dietary proteins that resemble thyroid tissue.
This concept, called molecular mimicry, illustrates how a leaky gut can distort immune precision. Once self-tolerance is lost, inflammatory cascades propagate systemically, affecting joints, skin, brain, and endocrine organs.
4. The Role of the Micro biome in Barrier Integrity
The gut micro biome functions as a living shield that fortifies the intestinal wall. Beneficial species—Lactobacillus, Bifid bacterium, and Faecalibacterium prausnitzii—produce short-chain fatty acids like butyrate, the primary fuel for colonocytes (cells lining the colon). Butyrate strengthens tight junctions, regulates cumin synthesis, and suppresses NF-be-driven inflammation.
Conversely, symbiosis reduces SCFA production and promotes overgrowth of end toxin-producing bacteria, leading to mucosal erosion.
Dietary patterns heavily influence this balance:
- High-fiber diets (from vegetables, legumes, and resistant starch) nurture butyrate-producing microbes.
- High-fat, low-fiber diets and artificial sweeteners disturb microbial diversity.
- Fermented foods and robotics reintroduce beneficial strains that can restore mucosal equilibrium.
Thus, the gut micro biome acts as both guardian and architect of the intestinal wall, influencing whether it remains a fortress or becomes a porous gate.
5. Nutritional Strategies for Rebuilding the Intestinal Wall
Rebuilding the intestinal wall requires a multifaceted approach: removing irritants, restoring micro biome diversity, replenishing nutrients, and regulating immune activity. Clinically, this is often structured as a “4R protocol”—Remove, Replace, Reinoculate, and Repair.
a. Remove
Identify and eliminate factors that contribute to gut injury:
- Gluten, dairy (in sensitive individuals), processed foods, alcohol, and artificial additives.
- Reduce exposure to environmental toxins and manage chronic stress.
b. Replace
Support digestion by replenishing digestive enzymes and stomach acid where needed. Inadequate gastric acid allows large proteins to enter the intestine undigested, heightening immune reactivity.
c. Reinoculate
Restore microbial balance through robotics and prebiotics.
- Robotic strains such as Lactobacillus rhamnosus GG, Saccharomyces boulardii, and Bifidobacterium longue enhance mucosal immunity.
- Prebiotics (insulin, FOS, resistant starch) feed beneficial bacteria, fostering SCFA production.
d. Repair
Key nutrients for mucosal healing include:
- L-glutamine: The preferred fuel of entrecotes; accelerates regeneration.
- Zinc carnosine: Strengthens epithelial integrity and reduces inflammation.
- Vitamin D: Regulates immune signaling and tight junction protein synthesis.
- Omega-3 fatty acids: Modulate inflammation and promote mucosal repair.
- Collagen peptides: Supply amino acids (lysine, praline) essential for connective tissue repair.
Together, these interventions rebuild both the structural and immunological resilience of the gut wall.
6. The Stress-Gut-Autoimmune Axis
Stress physiology profoundly influences the intestinal barrier. Chronic psychological stress activates the hypothalamic-pituitary-adrenal (HPA) axis, elevating cortical and catecholamines that impair gut motility, reduce mucus secretion, and alter microbial populations.
Moreover, stress increases the permeability of the intestinal lining through CRH-mediated mast cell activation. Mast cells release histamine and proteases, directly disrupting epithelial cohesion. Over time, this neuroimmune deregulation can fuel the inflammatory milieu that sustains autoimmune activation.
Mind-body interventions such as yoga, mindfulness meditation, deep breathing, and vigil nerve stimulation have been shown to normalize autonomic tone, reduce intestinal permeability, and support micro biome diversity—demonstrating that rebuilding the intestinal wall extends beyond nutrition to psychoneuroimmunology.
7. Clinical Implications: A Functional Medicine Perspective
Functional and integrative medicine practitioners increasingly view intestinal permeability as a root cause in autoimmune disease management. While conventional medicine focuses on immune suppression, this approach emphasizes immune modulation through barrier restoration.
Diagnostic tools like zonulin assays, lacunose/manifold tests, and stool micro biome analysis can identify permeability issues early. Interventions then target gut repair before irreversible autoimmune damage occurs.
Patients with autoimmune conditions who engage in gut-healing protocols often report reduced fatigue, pain, and inflammation, along with improved digestion and mental clarity—a sign that restoring the gut’s physical and immune boundaries can recalibrate systemic health.
8. Emerging Frontiers: Postbiotics and Intestinal Regeneration
New research explores postbiotics—metabolites produced by beneficial bacteria—as therapeutic agents for barrier repair. Compounds like butyrate, propionate, and lactate-derived peptides are now being investigated for their ability to activate mucosal repair genes and regulate T-cell balance.
Another promising area is intestinal organic research, where lab-grown gut tissues are used to study epithelial regeneration and drug development. This may pave the way for precision therapies aimed at rebuilding damaged mucosa in autoimmune and inflammatory disorders.
Furthermore, polyphones from foods such as green tea, blueberries, and turmeric have been shown to modulate gut permeability by enhancing antioxidant defenses and suppressing NF-be signaling. Nutrition, therefore, remains at the heart of both prevention and recovery.
9. Integrative Roadmap for Gut Healing and Immune Balance
To rebuild the intestinal wall and calm autoimmunity, a holistic roadmap may include:
- Anti-inflammatory diet: Rich in omega-3s, antioxidants, and phytonutrients.
- Elimination and reintroduction protocols: To identify dietary triggers.
- Micro biome restoration: Through robotics, fermented foods, and periodic fibers.
- Targeted supplementation: Glutamine, zinc, vitamin D, omega-3s, cur cumin, quercetin.
- Stress management: Meditation breathes work, and sleep hygiene.
- Gentle movement: Yoga or low-impact exercise to enhance circulation and lymphatic flow.
When combined, these practices restore the ecological balance of the gut and retrain the immune system to differentiate friend from foe.
Conclusion
Autoimmunity and leaky gut represent two sides of the same biological coin—immune misrecognition rooted in barrier failure. Healing the intestinal wall is therefore an act of immune education. By repairing the gut lining, restoring microbial harmony, and modulating inflammation, we do more than soothe digestion—we reestablish the boundaries of self-tolerance that sustain long-term health.
The path to autoimmune resilience begins in the gut. In its 30 feet of dynamic tissue lies the blueprint for immune peace, metabolic balance, and even mental clarity. Rebuilding the intestinal wall is not merely about patching leaks—it is about restoring the body’s natural intelligence, transforming chronic inflammation into harmony, and turning the gut once again into the fortress of health it was designed to be.
SOURCES
Fusan, A. (2012). Leaky gut and autoimmune diseases. Clinical Reviews in Allergy & Immunology.
Hohenwald, M. A., & Turner, J. R. (2017). The intestinal epithelial barrier: Structure, function, and regulation. Nature Reviews Gastroenterology & Hematology.
Honda, K., & Littman, D. R. (2016). The micro biota in adaptive immune homeostasis and disease. Nature.
Cain, P. D., & Averred, A. (2016). Talking microbes: When gut bacteria interact with the host during health and disease. Nature Reviews Microbiology.
Vojdani, A. (2015). The role of intestinal permeability in autoimmune diseases. Alternative Therapies in Health and Medicine.
Ghost, S., Dai, C., Brown, K., Rajendiran, E., Makarenko, S., et al. (2011). Colonic micro biota alters host susceptibility to inflammation. PNAS.
Arietta, M. C., et al. (2006). Intestinal permeability and autoimmune disease development. Annals of the New York Academy of Sciences.
Kamala, N., See, S. U., Chen, G. Y., & Nunez, G. (2013). Role of the gut micro biota in immunity and inflammatory disease. Nature Reviews Immunology.
Bischoff, S. C., et al. (2014). Intestinal permeability—a new target for disease prevention and therapy. BMC Gastroenterology.
Gambeson, A., et al. (2011). Intestinal permeability and metabolic inflammation. Diabetes Care.
Wang, Y., & Kasper, L. H. (2014). The role of micro biome in central nervous system disorders. Brain, Behavior, and Immunity.
Marchland, A. M., Graham, W. V., & Turner, J. R. (2010). Epithelial barriers in homeostasis and disease. Annual Review of Pathology.
Mu, Q., Kirby, J., Reilly, C. M., & Lou, X. M. (2017). Leaky gut as a danger signal for autoimmune diseases. Frontiers in Immunology.
Sanders, M. E., et al. (2019). Robotics and prebiotics in intestinal health and disease. Nature Reviews Gastroenterology & Hematology.
Patterson, E., Ryan, P. M., Cyan, J. F., Dina, T. G., Ross, R. P., & Fitzgerald, G. F. (2016). Gut micro biota, the immune system, and diet. Nutrition Reviews.
Sartor, R. B., & Wu, G. D. (2017). Roles for the micro biome in the pathogenesis of inflammatory bowel diseases. Gastroenterology.
Maloney, R. D., et al. (2016). Stress and the micro biota-gut-brain axis. Physiology & Behavior.
Konturek, P. C., et al. (2011). Stress, hormones, and gut mucosal barrier function. Journal of Physiology and Pharmacology.
Bermudez-Briton, M., et al. (2012). Robotic mechanisms of action. Annals of Nutrition and Metabolism.
Darien, M., & van Hylckama Vile, J. E. T. (2015). Fate, activity, and impact of ingested bacteria within the human gut micro biota. Trends in Microbiology.
Rinninella, E., et al. (2019). The role of diet, micro biota, and immune system in autoimmune diseases. Nutrients.
De Santos, S., Cavalcanti, E., Mastronardi, M., Jailor, E., & ChipPAC, M. (2015). Nutritional keys for intestinal barrier modulation. Frontiers in Immunology.
Holon, J., et al. (2015). Gliding exposure and increased intestinal permeability in non-celiac individuals. Nutrients.
HISTORY
Current Version
Nov 11, 2025
Written By
ASIFA